Gaseous StateHard
Question
28. Gold has a face centered cubic lattice with an edge length of the unit cube of 407 pm. Assuming the closest packing the diameter of the gold atom is
Options
A.576.6 pm
B.287.8 pm
C.352.5 pm
D.704.9 pm
Solution
For fcc lattice,
4 r = diagonal of the face = a√2
or 2r = diameter
=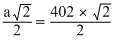 = 287.8pm
= 287.8pm
4 r = diagonal of the face = a√2
or 2r = diameter
=
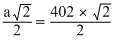 = 287.8pm
= 287.8pmCreate a free account to view solution
View Solution FreeMore Gaseous State Questions
′a′ and ′b′ are van der Waals′ constants for gases. Chlorine is more easily liquefied than...Dominance of storage repulsive forces among the molecules of the ges (Z = compressibility factor):...Value of gas constant R is...If V0 is the volume of a given mass of gas at 273 K at constant pressure, then according to Charle′s law, the volu...I, II, III are three isotherms respectively at T1, T2 and T3 as shown in graph. Temperature will be in order...