Gaseous StateHard
Question
I, II, III are three isotherms respectively at T1, T2 and T3 as shown in graph. Temperature will be in order
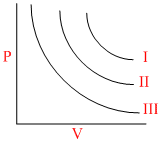

Options
A.T1 = T2 = T3
B.T1 < T2 < T3
C.T1 > T2 > T3
D.T1 > T2 = T3
More Gaseous State Questions
In Vander Waals equation of state of the gas law, the constant ′b′ is a measure of...A gaseous mixture containing He, CH4 & SO2 was allowed to effuse through a fine hole then find what molar ratio of gases...An element (atomic mass 100g/mol) having BCC structure has unit cell edge 400 pm. the density of element is (No. of atom...50 mL of each gas A and of gas B takes 200 and 150 s respectively for effusing through a pin hole under the similar cond...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...