Gaseous StateHard
Question
′a′ and ′b′ are van der Waals′ constants for gases. Chlorine is more easily liquefied than ethane because
Options
A.a and b for Cl2 < a and b for C2 H6
B.a for Cl2 < a for C2 H6 but b for 2 Cl > b for C2 H6
C.a for Cl2 > a for C2 H6 but b for 2 Cl < b for C2 H6
D.a and b for Cl2 > a and b for C2 H6
Solution
ease of liquefaction ∝ 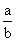
for ethane a = 5.49, b = 0.0638
for Cl2 a = 6.49, b = 0.0562
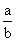
for ethane a = 5.49, b = 0.0638
for Cl2 a = 6.49, b = 0.0562
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Gaseous N2O4 dissociates into gaseous NO2 according to the reaction N2O4(g) ⇋ 2NO2(g) At 300 K and 1 atm pressure,...Just below this temperature, gases would theoretically have a negative volume (which is however not possible)-...Which nobal gas has maximum solubility in water :-...In the analysis of basic radicals, the group reagent H2S gas is generally used in the group:-...If most probable velocity is represented by ′a′ and fraction possessing it by ′f′, then with inc...