Gaseous StateHard
Question
Dominance of storage repulsive forces among the molecules of the ges (Z = compressibility factor):
Options
A.Depends on Z and indicated by Z = 1
B.Depends on Z and indicated by Z > 1
C.Depends on Z and indicated by Z < 1
D.Is independent of Z.
Solution
Repulsive force factor will decrease the compressibility factor i.e. so, value of Z> las
Z =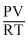
Due to repulsion value of PV will be greater than RT so Z > 1.
Z =
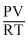
Due to repulsion value of PV will be greater than RT so Z > 1.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
As the temperature is raised from 20oC to 40oC, the average kinetic energy of neon atoms changes by a factor of which of...Which of the following compounds show sulimation ?...At low pressures (For 1 mole), the Vander Waal′s equation is written as V = RTThe compressibility factor is then e...Equal masses of H2, O2 and methane have been taken in a container of volume V at temperature 27oC in identical condition...Which of the following combination will produce H2 gas?...