JEE Advanced | 2015Gaseous StateHard
Question
The pair(s) of ions where BOTH the ions are precipitated upon passing H2S gas in presence of dilute HCl, is(are)
Options
A.Ba2+, Zn2+
B.Bi3+, Fe3+
C.Cu2+, Pb2+
D.Hg2+, Bi3+
Solution
(A) Ba+2 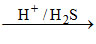 No ppt [because BaS is soluble in water]
No ppt [because BaS is soluble in water]
Zn+2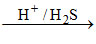 No ppt [because Zn+2 form ZnS in ammonical solution (IV group)
No ppt [because Zn+2 form ZnS in ammonical solution (IV group)
(large Ksp of ZnS)]
(B) Bi3+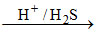 Bi2S3 ↓ [Bi3+ is II group cation
Bi2S3 ↓ [Bi3+ is II group cation
Brown/black ppt
Fe+3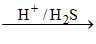 Fe+2 + S [because in acidic solution Fe+3 show redox reaction with H2S]
Fe+2 + S [because in acidic solution Fe+3 show redox reaction with H2S]
(C) Cu+2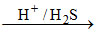 CuS↓ [Cu+2 is II group cation]
CuS↓ [Cu+2 is II group cation]
black ppt
Pb2+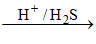 PbS↓ [Pb2+ is also II group cation]
PbS↓ [Pb2+ is also II group cation]
black ppt
(D) Hg2+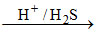 HgS↓ [Hg2+ is II group cation]
HgS↓ [Hg2+ is II group cation]
black ppt
Bi3+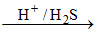 Bi2S3↓ [Bi3+ II group cation]
Bi2S3↓ [Bi3+ II group cation]
black/brown ppt
Zn+2
(large Ksp of ZnS)]
(B) Bi3+
Brown/black ppt
Fe+3
(C) Cu+2
black ppt
Pb2+
black ppt
(D) Hg2+
black ppt
Bi3+
black/brown ppt
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
For the diffusion of a gas at pressure P, the rate of diffusion is expressed by:...Three footballs are respectively filled with nitrogen, hydrogen and helium. If the leaking of the gas occurs with time f...Critical constant for a real gas are given asTC = 180 K ; Vc = 0.1231 L/molePC = 45 atm ;The correct statement for the r...See the figure-1 : The valves of X and Y are opened simultaneously. The white fumes of NH4Cl will first form at:...A mixture of hydrogen and oxygen at one bar pressure contains 20% by weight of hydrogen. Partial pressure of hydrogen wi...