Gaseous StateHard
Question
A gaseous mixture containing He, CH4 & SO2 was allowed to effuse through a fine hole then find what molar ratio of gases paning out initially ? If the mixture contains He, CH4 & SO2 in 1 : 2 : 3 mole ratio :-
Options
A.2 : 2 : 3
B.6 : 6 : 1
C.√2 : √2 : 3
D.8 : 4 : 3
Solution
r ∝ 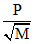
∴ nHe : nCH4 : nSO2 =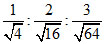
=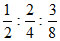 = 8 : 4 : 3
= 8 : 4 : 3
∴ nHe : nCH4 : nSO2 =
=
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
In vander Waal′s equation of state for a non ideal gas the term that accounts for intermolecular forces is :...Which of the following is not the correct set of pressure and volume at constant temperature and constant moles of gas ?...In the figure-2 the pressure of the confined gas will be...20 l of SO2 diffuses through a porous partition in 60 seconds. Volume of O2 diffuse under similar conditions in 30 secon...For real gases van der Waals equation is written as (V - nb) = nRT where ′a′ and ′b′ are van der...