Gaseous StateHard
Question
Helium atom is two times heavier than a hydrogen molecule at 298 k, the average kinetic energy of helium is :
Options
A.two times that of hydrogen molecule
B.same as that of the hydrogen molecule
C.four times that of a hydrogen molecule
D.half that of a hydrogen molecule
Solution
Average K.E. = 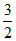 RT and T is constant 298 K
RT and T is constant 298 K
∴ K.E. is same for all gases at same Temperature.
∴ K.E. is same for all gases at same Temperature.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
At what temperature will the total KE of 0.3 mol of He be the same as the total KE of 0.40 mol of Ar at 400 K ?...In the analysis of basic radicals, the group reagent H2S gas is generally used in the group:-...Two flasks of equal volume are connected by a narrow tube (of negligible volume) all at 27oC and contain 0.70 moles of H...The volume temperature graphs of a given mass of an ideal gases constant pressure are shown below. What is the coorect o...A gaseous alkane is exploded with oxygen. The volume of O2 for complete combustion to CO2 formed is in the ratio 7/4. Th...