Chemical Kinetics and Nuclear ChemistryHard
Question
Bi210 has a half - life of 5days. The time taken, for 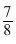 of a sample to decay, is :
of a sample to decay, is :
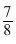 of a sample to decay, is :
of a sample to decay, is :Options
A.15 days
B.20 days
C.10 days
D.3.4 days
Solution
Half life of Bi210 = 5 days
∴ k =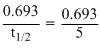 day-1
day-1
Using k =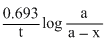
(where a = a0, (let) ⇒ x =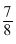 a0 t is time taken in dwcay and k is rate constant)
a0 t is time taken in dwcay and k is rate constant)
We get, t =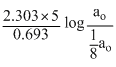
=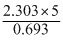 log 8 = 15 days
log 8 = 15 days
∴ k =
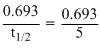 day-1
day-1Using k =
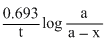
(where a = a0, (let) ⇒ x =
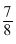 a0 t is time taken in dwcay and k is rate constant)
a0 t is time taken in dwcay and k is rate constant)We get, t =
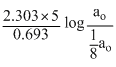
=
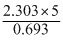 log 8 = 15 days
log 8 = 15 daysCreate a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the first-order reaction Tav (average life), T50 and T75 in the increasing order are :...The increasing order of basicity of the following compounds is : (A)Â Â Â Â Â (B)Â Â (C)Â Â Â Â Â Â (D)Â...Which is not true for a second order reaction ?...The rate equation for the reaction 2A + B → C is found to be: rate k[A][B]. The correct statement in relation to t...For the reaction A + B → C + D, the rate of the reaction may be expressed as r = . The negative sign in the above ...