Chemical Kinetics and Nuclear ChemistryHard
Question
For the reaction A + B → C + D, the rate of the reaction may be expressed as r = 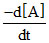 . The negative sign in the above expression indicates that
. The negative sign in the above expression indicates that
Options
A.Rate of the reaction is negative
B.Conc. of A is decreasing with time
C.Conc. of A is increasing with time
D.None of these
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For The Reaction, N2O5(g) → 2NO2(g) + 1/2 O2(g) the value of rate of disappearance of N2O5 is given as 6.25 ×...Among the following compounds (I-III), the correct order of reactivity towards elector Philip substitution reaction is...For a reaction 2NH3 → N2 + 3H2, it is observed that $- \frac{d\left\lbrack NH_{3} \right\rbrack}{dt} = K_{1}\left\lbrack...For which of the following reactions K310/K300 would be maximum ?...The correct increasing order of basic strength for the following compounds is :...