Atomic StructureHard
Question
The de-Broglie wavelength of an electron in the ground state of hydrogen atom is :
[K.E. = 13.6eV; leV = 602 × 10-19J]
[K.E. = 13.6eV; leV = 602 × 10-19J]
Options
A.33.28 nm
B.3.328 nm
C.0.3328 nm
D.0.0332 nm
Solution
We know that
K.E. =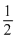 mv2
mv2
∴ v =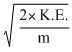
=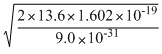
2.18824 × 106m/s
No since, λ =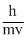
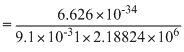
= 0.3328 × 10-9
= 0.3328 nm
K.E. =
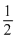 mv2
mv2∴ v =
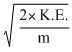
=
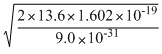
2.18824 × 106m/s
No since, λ =
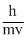
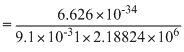
= 0.3328 × 10-9
= 0.3328 nm
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Total no. of lines in Lyman series of H spectrum will be (where n = no. of orbits)...Beryllium fourth electron will have the four quantum numbers :-...The correct statement(s) about Cr2+ and Mn3+ is (are)[Atomic numbers of Cr = 24 and Mn = 25]...Which of the following statements is/are correct?In the reaction xCu3P + y Cr2O72- Cu2+ + H3PO4 + Cr3+...Two electromagnetic radiations have wave numbers in the ratio 2: 3. Their energies per quanta will be in the ratio...