Atomic StructureHard
Question
The correct statement(s) about Cr2+ and Mn3+ is (are)
[Atomic numbers of Cr = 24 and Mn = 25]
[Atomic numbers of Cr = 24 and Mn = 25]
Options
A.Cr2+ is a reducing agent
B.Mn3+ is an oxidizing agent
C.Both Cr2+ and Mn3+ exhibit d4 electronic configuration
D.When Cr2+ is used as a reducing agent, the chromium ion attains d5 electronic configuration
Solution
(A) Cr+2 is a reducing agent : CORRECT STATEMENT
Cr+2 is a reducing agent as its configuration changes from d4 to d3 and Cr+3 having a half filled t2g level in presence of H2O
Cr+2 → Cr+3 + e−
3d44so 3d34so
(B) Mn+3 is a oxidizing agent : CORRECT STATEMENT
The change from Mn+3 to Mn+2 results in the half filled (d5) configuration which has extra stability
Mn+3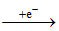 Mn+2
Mn+2
3d44so 3d54so
[extra stable half filled (d5) configuration]
(C) Both Cr+2 and Mn+3 exhibit d4 electronic configuration : CORRECT STATEMENT
Cr+2 → 3d44so
Mn+3 → 3d44so
(D) When Cr+2 is used as reducing agent chromium ion attain d5 electronic configuration : INCORECT
Cr+2 → Cr+3 + e−
3d44so 3d34so
Reducing agent It attain d3 electronic configuration
Cr+2 is a reducing agent as its configuration changes from d4 to d3 and Cr+3 having a half filled t2g level in presence of H2O
Cr+2 → Cr+3 + e−
3d44so 3d34so
(B) Mn+3 is a oxidizing agent : CORRECT STATEMENT
The change from Mn+3 to Mn+2 results in the half filled (d5) configuration which has extra stability
Mn+3
3d44so 3d54so
[extra stable half filled (d5) configuration]
(C) Both Cr+2 and Mn+3 exhibit d4 electronic configuration : CORRECT STATEMENT
Cr+2 → 3d44so
Mn+3 → 3d44so
(D) When Cr+2 is used as reducing agent chromium ion attain d5 electronic configuration : INCORECT
Cr+2 → Cr+3 + e−
3d44so 3d34so
Reducing agent It attain d3 electronic configuration
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The excitation energy of an electron from second orbit to third orbit of a hydrogen-like atom or ion with +Ze nuclear ch...In the absence of external magnetic field, d-orbital is...From the α-particle scattering experiment, Rutherford concluded that...Ground state electronic configuration of nitrogen atom can be represented by :...A photo sensitive surface is receiving light of wavelength 5000 Å at the rate of 10−7 J/s. The number of photons receive...