SolutionHard
Question
At 300K, the vapour pressure of an ideal solution containing 3 mole of A and 2 mole of B is 600 torr. At the same temperature if 1.5 mole of A and 0.5 mole of C (non-volatile) are added to this solution the vapour pressure of solution in creases by 30 torr. What is the value of p0B ?
Options
A.940
B.405
C.90
D.none of these
Solution
P = p0AxA + p0BxB
600 = p0A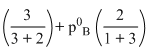
3p0A + 2p0B = 3000
630 = p0A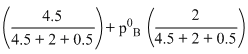
4.5p0A + 2p0B = 4410
1.5p0A = 1410; p0A = 940 and p0B = 90
600 = p0A
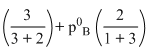
3p0A + 2p0B = 3000
630 = p0A
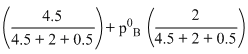
4.5p0A + 2p0B = 4410
1.5p0A = 1410; p0A = 940 and p0B = 90
Create a free account to view solution
View Solution FreeMore Solution Questions
To an acid solution of an anion, a few drops of KMnO4 solution are added which of the following, if present will not dec...Which of the following liquid pairs shows a positive deviation from Raoult′s law ?...When during electrolysis of a solution of AgNO3 9650 coulombs of charge pass through the electroplating bath, the mass o...In the above problem, if the solution is cooled to −0.200°C, then how many grams of ice would separate?...At 323 K, the vapour pressure in millimeters of mercury of a methanol-ethanol solution is represented by the equation p ...