SolutionHard
Question
At 323 K, the vapour pressure in millimeters of mercury of a methanol-ethanol solution is represented by the equation p = 120 XA + 140, where XA is the mole fraction of methanol. Then the value of 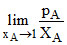 is
is
Options
A.250 mm
B.140 mm
C.260 mm
D.20 mm
Solution
As XA → 1. Then we will have pure methanol so PT = 120 XA + 140 = 120 + 140 = 260 mm of Hg.
Create a free account to view solution
View Solution FreeMore Solution Questions
At 35oC, the vapour pressure of CS2 is 512 mmHg, and of acetone is 344 mmHg. A solution of CS2 and acetone in, which the...When 2 g non-volatile hydrocarbon containing 94.4% carbon by mass is dissolved in 100 g of benzene, the vapour pressure ...If P0 and PS, are the V.P. of solvent and its solution respectively and N1 and N2 are the mole fraction of solvent and s...A graph plotted between vs , d (where P is osmotic pressure of solution of a solute of mol. wt. m and d is its density a...On increasing the altitude at constant temperature, the vapour pressure of a liquid...