SolutionHard
Question
0.450 g of urea (mol.wt. 60) in 22.5 g of water show 0.170oC of elevation in boiling point. The molal elevation constant of water is :
Options
A.0.051oC
B.0.51oC
C.5.1oC
D.0.83oC
More Solution Questions
Freezing point of an aqueous solution is (-0.186)oC. Elevation of boiling point of the same solution is Kb = 0.512 0C, K...Mole fraction of the toluene in the vapour phase which is in equilibrium with a solution of benzene (po = 120 Torr) and ...Total vapour pressure of mixture of 1 mol of volatile component A (pAo = 100 mmHg) and 3 mol of volatile component B (pB...A Solution of sucrose (molar mass = 342 gm-mol-1) has been prepared by dissolving 68.5 g of sucrose in 1000 g of water. ...Which of the following is a buffer solution ?...
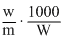
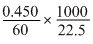
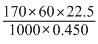 = 0.51oC
= 0.51oC