SolutionHard
Question
Mole fraction of the toluene in the vapour phase which is in equilibrium with a solution of benzene (po = 120 Torr) and toluene (po = 80 Torr) having 2.0 mol of each is
Options
A.0.50
B.0.25
C.0.60
D.0.40
Solution
PT = XA poA + XB poB
=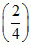 × 80 +
× 80 + 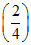 × 120 = 100 Torr
× 120 = 100 Torr
Now mole fraction in vapour phase =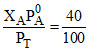 = 0.4.
= 0.4.
=
Now mole fraction in vapour phase =
Create a free account to view solution
View Solution FreeMore Solution Questions
What is the OH- concentration of an aqueous solution of a substance whose pH = 3.2 :-...Which one of the following graphs accurately represents the plot of partial pressure of ${CS}_{2}$ vs its mole fraction ...Water should boil at 90°C at pressure...Which of the following is less than zero for ideal solutions ?...A glucose solution is isopiestic with blood. What is the osmolarity of glucose solution?...