ElectrochemistryHard
Question
During electrolysis of water the volume of O2 Liberated is 2.24 dm3. The volume of hydrogen liberated, under same condition will be
Options
A.2.24dm3
B.1.12 dm3
C.4.48 dm3
D.0.56 dm3
Solution
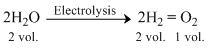
Thus, the volume of hydrogen librated is twice that volume of oxygen liberated. When2.24dm3 of oxygen is librated the volume of hydrogen librated will be 2 × 2.4dm3 or 4.48dm3
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The solubility product of silver iodide is 8.3 × 10-17 and the standard reduction potential of Ag, Ag+ electrode is...Cost of electricity for the production of ′X′ litre H2 at NTP at cathode is Rs. X, then cost of electricity ...Given that EoK+/K = - 2.93V; EoFe2+/Fe = - 0.44V ; EoZn2+/Zn = - 0.76V ; EoC+2/Cu = 0.34V; Based on this Data, which of ...A current of 9.65 ampere is passed through the aqueous solution NaCl using suitable electrodes for 1000 s. The amount of...Equivalent conductance of an electrolyte containing NaF at infinite dilution is 90.1 Ohm-1 cm2 If NaF is replaced by KF ...