ElectrochemistryHard
Question
A current of 9.65 ampere is passed through the aqueous solution NaCl using suitable electrodes for 1000 s. The amount of NaOH formed during electrolysis is
Options
A.2.0 g
B.4.0 g
C.6.0 g
D.8.0 g
Solution
2H2O + 2e- → H2 + 2OH-
No. of Faraday passed =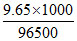 = 0.1 F
= 0.1 F
nOH- formed = 0.1 mol
nNaOH = 0.1 mol ≡ 4 gm.
No. of Faraday passed =
nOH- formed = 0.1 mol
nNaOH = 0.1 mol ≡ 4 gm.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
In which of the following molecules π - electron density in ring is maximum :...An unknown alcohol is treated with the ″Lucasreagent′ to determine whether the alcohol is primary, secondary...Sulfide ores are common for the metals -...In a galvanic cell, the salt bridge...Assertion :- BeSO4 is more soluble in water than BaSO4.Reason :- In BeSO4 H.E. exceeds its L.E....