Mole ConceptHard
Question
1021 molecules are removed from 200mg of CO2. The moles of CO2 left are :
Options
A.2.88 × 10-3
B.28.8 × 10-3
C.288 × 10-3
D.28.8 × 103
Solution
No. of moles = 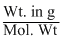
no. of moles in 200mg =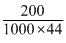 = 4.5 × 10-3 moles
= 4.5 × 10-3 moles
No.of moles in 1021 molecules
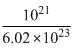 = 1.67 × 10-3 moles
= 1.67 × 10-3 moles
No. of moles left = (4.5 - 1.67) × 10-3
= 2.88 × 10-3
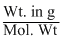
no. of moles in 200mg =
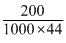 = 4.5 × 10-3 moles
= 4.5 × 10-3 molesNo.of moles in 1021 molecules
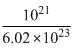 = 1.67 × 10-3 moles
= 1.67 × 10-3 molesNo. of moles left = (4.5 - 1.67) × 10-3
= 2.88 × 10-3
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
A mixture of ethylene and excess of H2 had a pressure of 600 mm Hg. The mixture was passed over nickel catalyst to conve...If an iodized salt contains 1% of KI and a person takes 2 g of the salt every day, the iodine ions going into his body e...An amount of 1.0 × 10–3 moles of Ag+ and 1.0 × 10–3 moles of CrO4 2− reacts together to form solid Ag2CrO4. What is the ...A volume of 200 ml of oxygen is added to 100 ml of a mixture containing CS2 vapour and CO, and the total mixture is burn...If the yield of chloroform obtainable from acetone and bleaching powder is 75%, then what mass of acetone is required fo...