Mole ConceptHard
Question
A mixture of ethylene and excess of H2 had a pressure of 600 mm Hg. The mixture was passed over nickel catalyst to convert ethylene to ethane. The pressure of the resultant mixture at the similar conditions of temperature and volume dropped to 400 mm Hg. The fraction of C2H4 by volume in the original mixture is
Options
A.1/3 rd of the total volume
B.1/4 th of the total volume
C.2/3 rd of the total volume
D.1/2 of the total volume
Solution
Let there be n mole of (C2H4 + H2)
Mole of C2H4 = x
H2 = (n - x) mole
C2H4 + H2 → C2H6
Mole of C2H4 = x
H2 = (n - x) mole
C2H4 + H2 → C2H6
x n-x
0 n - 2x x mol
After reaction (C2H6 + H2 left)
x + n - x - x = n - x
[Total H2 = (n - x), H2 reacted = x]
H2 left = (n - x - x)
n ∝ 600; n - x ∝ 400
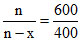 ;
;
0 n - 2x x mol
After reaction (C2H6 + H2 left)
x + n - x - x = n - x
[Total H2 = (n - x), H2 reacted = x]
H2 left = (n - x - x)
n ∝ 600; n - x ∝ 400
x/n = 1/3 = fraction of C2H4 by volume
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The strength of 10-2 M Na2CO3 solution in terms of molality will be (density of solution = 1.10 g mL-1). (Molecular weig...Density of ozone relative to oxygen is under the same temperature and pressure...How many grams of 90% pure Na2SO4 can be produced from 250 g of 95% pure NaCl?...A sample of impure cuprous oxide contains 66.67% copper by mass. What is the percentage of pure Cu2O in the sample? (Cu ...Accumulation of which of the following molecules in the muscles occurs as a result of vigorous exercise ?...