Chemical EquilibriumHard
Question
For the following mechanism, P + Q 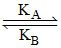 PQ
PQ 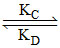 R at equilibrium
R at equilibrium 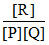 is :
is :
[k represents rate constant]
[k represents rate constant]
Options
A.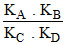
B.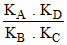
C.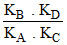
D.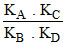
Solution
On multiply equation (i) and (ii) we get
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
An amount of solid NH4HS is placed in a flask already containing ammonia gas at a certain temperature and 0.50 atm. Pres...I2 + I−$\rightleftharpoons$I3−. This reaction is set up in aqueous medium. We start with 1 mol of I2 and 0.5 mol of I¯ i...For the equilibrium at 298 K; N2O4(g) ⇋ 2NO2(g); G-N2O4 = 100 kJ mol-1 and G-NO2 = 50 kJ mol-1. If 5 mol of N2O4 a...The condition suitable for forming atomic chlorine from molecular chlorine is...A gaseous substance AB2(g) converts to AB(g) in the presence of solid A as AB2(g) + A(s)$\rightleftharpoons$ 2AB(g).The ...