Chemical EquilibriumHard
Question
1 mol of N2 is mixed with 3 mol of H2 in a litre container. If 50% of N2 is converted into ammonia by the reaction N2 + 3H2(g) ⇋ 2NH3(g), then the total number of moles of gas at the equilibrium are
Options
A.1.5
B.4.5
C.3.0
D.6.0
Solution
α = 50 % = 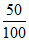 = 0.5
= 0.5
N2 + 3H2 ⇋ 2NH3
Initial 1 3 0
Final 1 - α 3 - 3α 2α
Total moles = 1 - α + 3 - 3α + 2α
= 4 - 2α
= 4 - 2 × 0.5 = 3
N2 + 3H2 ⇋ 2NH3
Initial 1 3 0
Final 1 - α 3 - 3α 2α
Total moles = 1 - α + 3 - 3α + 2α
= 4 - 2α
= 4 - 2 × 0.5 = 3
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For a reversible reaction $A\overset{\quad K_{1}K_{2}\quad}{\leftleftarrows}B$, the initial molar concentration of A and...For the given chemical equation 2X(g) + Y(g) $\rightleftharpoons$2Z(g) + 8 kcal, which combination of pressure and tempe...When calcium acetate is dissolved in water, heat is evolved. If the temperature is raised, then the solubility of calciu...In the system, LaCl3(s) + H2O(g) + heat ⇋ LaClO(s) + 2HCl(g), equilibrium is established. More water vapour is add...PCl5(g) $\rightleftharpoons$PCl3(g) + Cl2(g). In the above reaction, the partial pressure of PCl3, Cl2 and PCl5 are 0.3,...