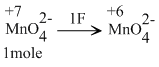AIPMT | 2014ElectrochemistryHard
Question
When 0.1 mol MnO42- is oxidised the quantity of electricity required to completely oxidise MnO42- to MnO4- is
Options
A.96500 C
B.2 × 96500 C
C.9650 C
D.96.50 C
More Electrochemistry Questions
Two platinum electrodes were immersed in a solution of CuSO4 and electric current was passed through the solution. After...Cu+ + e- → Cu, Eo = x1 volt ;Cu2+ + 2e- Cu, Eo = x2 volt, then forCu2+ + e- Cu+, Eo (volt) will be -...BY the electrolysis of aqueous solution of CuSO4, the products obtained at both the electrodes are...The elctrode potentials for Cu2+ (aq) + e- → Cu+(aq) and Cu+ (aq) + e- → Cu (s) are +0.15 V and +0.50 V resp...Molar conductance of 0.1 M acetic acid is 7 ohm-1 cm2 mol-1. If the molar cond. of acetic acid at infinite dilution is 3...