ElectrochemistryHard
Question
The elctrode potentials for
Cu2+ (aq) + e- → Cu+(aq)
and Cu+ (aq) + e- → Cu (s)
are +0.15 V and +0.50 V respectively. The value of EoCu2+/Cu will be
Cu2+ (aq) + e- → Cu+(aq)
and Cu+ (aq) + e- → Cu (s)
are +0.15 V and +0.50 V respectively. The value of EoCu2+/Cu will be
Options
A.0.325 V
B.0.650 V
C.0.150 V
D.0.500 V
Solution
Cu2+ + e- → Cu+
Eo1 = 0.15 V; ᐃGo1 = - n1Eo1F
Cu+ + e- → Cu; Eo2 = 0.50 V; ᐃGo2 = - n2Eo2F

Cu+ + e- → Cu ; Eo = ?; ᐃGo = - nEoF
ᐃGo = ᐃGo1 + ᐃGoo
or - 2EoF = - 2F × 0. 15 + ( - 1F × 0.15)
or - 2EoF = - 0.15F - 0.50F
or - 2 FEo = - F ( 0.15 + 0.50 )
∴ Eo =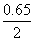 = 0.325 V
= 0.325 V
Eo1 = 0.15 V; ᐃGo1 = - n1Eo1F
Cu+ + e- → Cu; Eo2 = 0.50 V; ᐃGo2 = - n2Eo2F

Cu+ + e- → Cu ; Eo = ?; ᐃGo = - nEoF
ᐃGo = ᐃGo1 + ᐃGoo
or - 2EoF = - 2F × 0. 15 + ( - 1F × 0.15)
or - 2EoF = - 0.15F - 0.50F
or - 2 FEo = - F ( 0.15 + 0.50 )
∴ Eo =
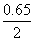 = 0.325 V
= 0.325 VCreate a free account to view solution
View Solution FreeMore Electrochemistry Questions
A solution containing Na+, NO3-, Cl-, and SO42- ions, all at unit concentrations, is electrolyzed between nickel anode a...Expermentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+and M3+ in its oxide. Fracti...Three faradays of electricity was passed through an aqueous solution of iron (II) bromide. The mass of iron metal (at. m...Red hot carbon will remove oxygen from the oxide AO and BO but not from MO, while B will remove oxygen from AO. The acti...If 0.224 L of H2 gas is formed at the cathode, the volume of O2 gas formed at the anode under identical conditions, is...