JEE Advanced | 2014Chemical Kinetics and Nuclear ChemistryHard
Question
For the elementary reaction M → N, the rate of disappearance of M increases by a factor of 8 upon doubling the concentration of M. The order of the reaction with respect to M is
Options
A.4
B.3
C.2
D.1
Solution
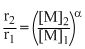
8 = (2)α
α = 3
So, order of reaction is 3.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The energy equivalent of 2.0 mg mass defect is...During the kinetic study of the reaction, 2A + B → C + D, following results were obtained Based on the above data ...For an endothermic reaction, energy of activation is Ea and enthalpy of reaction is ᐃH (both of these in KJ/mol). ...Anisotope is :...The inversion of cane sugar proceeds with half life of 500 min at pH 5 for any concentration of sugar. However, if pH = ...