Chemical Kinetics and Nuclear ChemistryHard
Question
The inversion of cane sugar proceeds with half life of 500 min at pH 5 for any concentration of sugar. However, if pH = 6, the half life change to 50 min. The rate law expression for the sugar inversion can be written as
Options
A.r = k[sugar]2[H]6
B.r = k[sugar]1[H]0
C.r = k[sugar]0[H+]6
D.r = k[sugar]0[H+]1
Solution
Sincet1/2 does not depends upon the sugar concentration means it is first order w.r.t.[sugar]
∴ t1/2 ∝ [sugar]1
t1/2 × an-1 = k
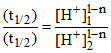
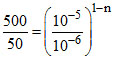
10 = (10)1-n ⇒ n = 0.
∴ t1/2 ∝ [sugar]1
t1/2 × an-1 = k
10 = (10)1-n ⇒ n = 0.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Among CaH2, BeH2, BaH2, the order of ionic character is...Consider an endothermic reaction, X → Y with the activation energies Eb and Ef for the backward and forward reacti...Among the following compounds (I-III), the correct order of reactivity towards elector Philip substitution reaction is...In the nuclear transmutation49Be + X → 48Be + Y (X, Y) is(are)...A second order reaction requires 70 min to change the concentration of reactants from 0.08 M to 0.01 M. How much time wi...