Atomic StructureHard
Question
What is the wavelength of the least energetic photon emitted in the Lyman series of the hydrogen spectrum ?
Options
A.150nm
B.122nm
C.102nm
D.82nm
Solution
The least energy is corresponding to longest wavelength,which is λ = 1216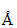 .
.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
For a given plate voltage, the plate current in a triode valve is maximum when the potential of...The transtion from the state n = 4 to n = 3 in a hydrogen like atom results in ultraviolet radiation. Infrared radiation...In hydrogen atom spectrum, ( $R \rightarrow$ Rydberg's constant)A. the maximum wavelength of the radiation of Lyman seri...Following are graphs of electric field variation with distance (r) for four different charge configurations.Choose the c...Calculate power output of reactor, If it takes 30 days to use up 2 kg of fuel, and if each fission gives 185 MeV of usea...