ThermodynamicsHard
Question
Two identical containers A and B with friction pistons contain the ideal gas at the same temperature and the same volume v. The mass of the gas in A is mA and b in mB. The gas in each cylinder is now allowed to expand isothermally to the same final volume 2v. The changes in pressure in a b are found to be ᐃp and 1.5 ᐃp respectively. Then
Options
A.4mA = 9mB
B.2mA = 3mB
C.3mA = 2mB
D.9mA = 4mB
Solution
The initial pressure in the two containers will be
PA =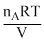 = mA
= mA 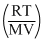
PB =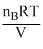 = mB
= mB 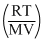
After isothermal expasion, pressure will be
P′B =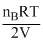 = mB
= mB 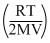
P′A =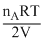 = mA
= mA 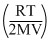
∴ - ᐃPA = PA - P′A = mA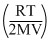
∴ - ᐃPB = PB - P′B = mB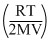
But ᐃPA = ᐃP and - ᐃPB = 1.5 ᐃ
So,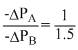 ⇒ - ᐃPA =
⇒ - ᐃPA = 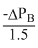
mA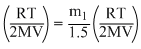
or mA =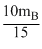
∴ 3mA = 2mB
PA =
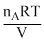 = mA
= mA 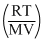
PB =
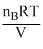 = mB
= mB 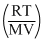
After isothermal expasion, pressure will be
P′B =
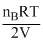 = mB
= mB 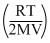
P′A =
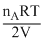 = mA
= mA 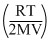
∴ - ᐃPA = PA - P′A = mA
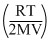
∴ - ᐃPB = PB - P′B = mB
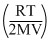
But ᐃPA = ᐃP and - ᐃPB = 1.5 ᐃ
So,
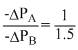 ⇒ - ᐃPA =
⇒ - ᐃPA = 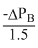
mA
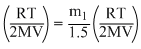
or mA =
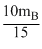
∴ 3mA = 2mB
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
70 cal of heat are required to raise the temperature of 2 moles of an ideal diatomic gas at constant pressure 30oC to 35...1 calorie is equal to how many joules ?...The figure shows the P-V plot of an ideal gas taken through a cycle ABCDA. The part ABC is a semi-circle and CDA is half...The volume of a 2 mole ideal gas at 27oC is V. It expands adiabatically to the volume 2V. If γ = 5/3, R = 8.31 J/mo...One kg of a diatomic gas is at a pressure of 8 × 104 N/m2. The density of the gas is 4 kg/m-3. What is the energy o...