ThermodynamicsHard
Question
One kg of a diatomic gas is at a pressure of 8 × 104 N/m2. The density of the gas is 4 kg/m-3. What is the energy of the gas due to its thermal motion?
Options
A.3 × 104J
B.5 × 104J
C.6 × 104J
D.7 × 104J
Solution
Thermal energy corresponds to internal energy
Mass = 1 kg
density = 8 kg/m3
⇒ Volume =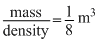
Pressure = 8 × 104 N/m2
∴ Internal Energy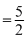 P × v = 5 × 104 J
P × v = 5 × 104 J
Mass = 1 kg
density = 8 kg/m3
⇒ Volume =
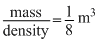
Pressure = 8 × 104 N/m2
∴ Internal Energy
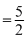 P × v = 5 × 104 J
P × v = 5 × 104 JCreate a free account to view solution
View Solution FreeMore Thermodynamics Questions
At constant pressure wokdone in changing the volume from V1 to V2 is :-...During the melting of a slab of ice at 273 K at atomospheric pressure...If the heat of 110 J is added to a gaseous system, change in internal energy is 40 J, then the amount of external work d...The density of a substance at 0oC is 10g / cc and at 100oC, its density is 9.7g/cc. The coefficient of linear expansion ...If the temperature of a black body increases from 7oC to 287oC then rate of energy radiation increases by...