ThermodynamicsHard
Question
70 cal of heat are required to raise the temperature of 2 moles of an ideal diatomic gas at constant pressure 30oC to 35oC. The amount of heat required (in calorie) to raise the temperature of the same gas through the same gas (30oC to 35oC) at constant volume is
Options
A.30
B.50
C.70
D.90
Solution
Q1 = nCp ᐃT, Q2 = nCv ᐃT, 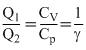
or Q2 =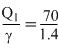 = 50 cal
= 50 cal
∴ Correct option is (b)
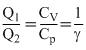
or Q2 =
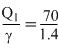 = 50 cal
= 50 cal∴ Correct option is (b)
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The real coefficient of volume expansion of glycerine is 0.000597 per oC and linear coefficient of expansion of glass is...A Carnot engine, whose efficiency is 40% takesin heat from a source maintained at atemperature of 500 K. It is desired t...In a water fall the water falls from a height of 100 m. If the entire energy of water is converted into heat. The rise i...If the temperature of a body is 140oF, its value in oC will be :-...The relation Cp − Cv = R/J implies :-(J−Joule constant)...