ThermodynamicsHard
Question
To rigid boxes containing different ideal gases are placed on a table. Box A contains one mole are placed on a table. Box A contains one mole of nitrogen at temperature T0, while box B contains one mole of helium at temperature 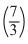 T0. The boxes are then put into thermal contect with each until the gases reach a common final temprature of the gases, Tf in terms of T0 is
T0. The boxes are then put into thermal contect with each until the gases reach a common final temprature of the gases, Tf in terms of T0 is
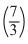 T0. The boxes are then put into thermal contect with each until the gases reach a common final temprature of the gases, Tf in terms of T0 is
T0. The boxes are then put into thermal contect with each until the gases reach a common final temprature of the gases, Tf in terms of T0 is Options
A.Tf = 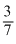
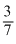
B.Tf = 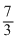
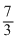
C.Tf = 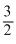
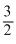
D.Tf = 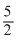
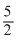
Solution
Heat lost by He = Heat gained by N2
n1Cv1ᐃT1 = n2Cv2 ᐃT2
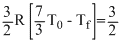 R[Tf - T0]
R[Tf - T0]
7T0 - 3Tf = 5Tf - 5T0
⇒ 12T0 = 8Tf ⇒ Tf =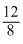 T0 ⇒ Tf =
T0 ⇒ Tf = 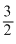 T0
T0
n1Cv1ᐃT1 = n2Cv2 ᐃT2
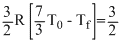 R[Tf - T0]
R[Tf - T0]7T0 - 3Tf = 5Tf - 5T0
⇒ 12T0 = 8Tf ⇒ Tf =
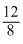 T0 ⇒ Tf =
T0 ⇒ Tf = 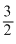 T0
T0Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A gas is taken through the cycle A → B → C → A, as shown, What is the net work done by the gas ?...When a system is taken from state i to state f along the path ′iaf′, it is found that Q = 50 cal and W = 20 ...In the given graph the isothermal curves are :-...The volume of a gas expands by 0.25 m3 at a constant pressure of 103 N/m2. The work done is equal to...N moles of a monoatomic gas is carried round the reversible rectangular cycle ABCDA as shown in the diagram. The tempera...