ThermodynamicsHard
Question
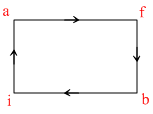
When a system is taken from state i to state f along the path ′iaf′, it is found that Q = 50 cal and W = 20 cal. If along the path ′ibf′ Q = 36 cal. W along the path ibf is

Options
A.6 cal
B.16 cal.
C.66 cal.
D.14 cal.
Solution
First law of thermodynamics ᐃU = ᐃQ − ᐃW
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Four curves A, B, C and D are drawn in the adjoining figure for a given amount of gas. The curves which represent adiaba...As shown in the diagram for a closed path ABCA :...An ideal gas heat engine operates in a cornot cycle between 227oC and 127oC. It absorbs 6 kcal at the higher temperature...One mole ideal gas is compressed adiabatically at 27oC. Its temperature becomes 102oC. The work done in this process wil...On mixing 1 gm mole of a monoatomic with 1 gm mole of a diatomic gas the specific heat of mixture at constant volume wil...