ThermodynamicsHard
Question
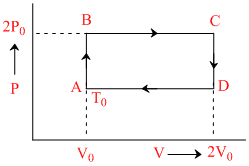
N moles of a monoatomic gas is carried round the reversible rectangular cycle ABCDA as shown in the diagram. The temperature at A is T0. The thermodynamic efficiency of the cycle is:

Options
A.15 %
B.50 %
C.20 %
D.25 %
Solution
Heat absorbed = Work done by gas at = 2P0 (2V0 - V0) = 2PV0V0
Net work done by the gas = Workdone by the gas - Workdone on the gas
= 2P0V0 - P0V0
So efficiency =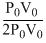 × 100 = 50%
× 100 = 50%
Net work done by the gas = Workdone by the gas - Workdone on the gas
= 2P0V0 - P0V0
So efficiency =
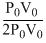 × 100 = 50%
× 100 = 50%Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
During the melting of a slab of ice at 273 K at atomospheric pressure...In an isothermal thermodynamical process, the value of work done by a system is...The amount of work done in converting 1 gm ice at -10o C into steam at 100o C will be : -...Three objects colored black, gray and white can withstand hostile conditions upto2800oC. These objects are thrown into a...The wavelength of maximum energy released during an atomic explosin was 2.93 × 10-10m. The maximum temperature atta...