NEET | 2013ThermodynamicsHard
Question
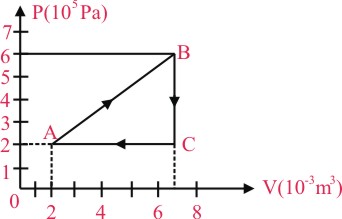
A gas is taken through the cycle A → B → C → A, as shown, What is the net work done by the gas ?

Options
A.- 2000 J
B.2000 J
C.1000 J
D.Zero
Solution
Net work done = Area of triangle ABC
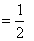 × [(72) × 10-3] [(6 - 2) × 105]
× [(72) × 10-3] [(6 - 2) × 105]
= 1000 J.
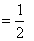 × [(72) × 10-3] [(6 - 2) × 105]
× [(72) × 10-3] [(6 - 2) × 105]= 1000 J.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
An amount Q of heat is added to a monoatomic ideal gas in thermodynamic process. In the process gas expands and does Q/2...A gas mixture contain 1 gm. H2 and 1 gm. He if temp. of gas mixture increase 0oC to 100o C at isobaric process. Than fin...A system is taken along the paths A and B as shown. If the amounts of heat given in these processes are ᐃQA and &#...For isometric process : -...A block of ice at -10oC is slowly heated and converted to steam at 100oC. Which of the following curves represents the p...