ThermodynamicsHard
Question
In an adiabatic change, the pressure and temperature of a monoatomic gas are related with relation as P ∝ TC, where C is equal to :
Options
A.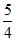
B.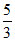
C.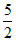
D.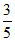
Solution
P = kTc ⇒ PT-c = k
For adiabatic change
PVγ = constant [where γ is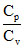 ]
]
or PTγ/1-γ = constant
Comparing it with given relation
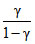 = - C
= - C
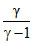 = C
= C
For monoatomic gas, γ =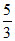
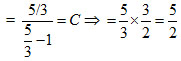
For adiabatic change
PVγ = constant [where γ is
or PTγ/1-γ = constant
Comparing it with given relation
For monoatomic gas, γ =
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Two moles of monoatomic gas are mixed with 1 mole of a diatomic gas. The γγ for the mixture is :...Water is used to cool the radiators of engines in cars because :...One mole of an ideal gas goes form an initial state A to final state B via two processes: It first undergoesisothermal e...An ideal monoatomic gas is taken round the cycle ACBCA as shown in the p-V diagram (see figure). The work done during th...One mole of a monatomic ideal gas is taken along two cyclic processes E → F → G → E and E → F &#...