ThermodynamicsHard
Question
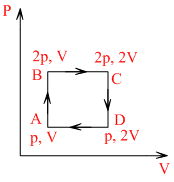
An ideal monoatomic gas is taken round the cycle ACBCA as shown in the p-V diagram (see figure). The work done during the cycle is

Options
A.pV
B.2pV
C.1/pV
D.zero
Solution
Work done in cyclic process = area between the cycle
= AB × BC = (2 p - p) × (2V - V ) = pV
∴ Correct option is (a).
= AB × BC = (2 p - p) × (2V - V ) = pV
∴ Correct option is (a).
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Which of the following is incorrect regarding the first law of thermodynamics?...The radiation from the sun, Incident normally at the surdace of the earth is 20 kcal/m2 min.What would have the radiant ...A monoatomic gas at a pressure P, having a volume V expands isothermally to a volume 2 V and then adiabatically to a vol...When a system is taken from state i to state f along the path ′iaf′, it is found that Q = 50 cal and W = 20 ...An ideal gas at 27oC is compressed adiabatically to its original volume [TVγ-1] and , Then the rise in temprature w...