ThermodynamicsHard
Question
One mole of an ideal gas goes form an initial state A to final state B via two processes: It first undergoesisothermal expansion from volume V to 3V and then its volume is reduced from 3V to V at constantpressure. The correct P-V diagram representing the two processes is:
Options
A.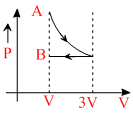

B.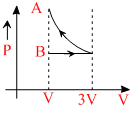

C.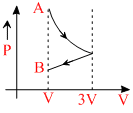

D.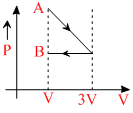

More Thermodynamics Questions
One mole of an ideal gas follows the process P = , P0 and V0 are constants when the volume of gas is changed from V0 to ...In a process on a closed system of ideal gas, the initial pressure and volume is equal to the final pressure and volume...A sample of gas expands from volume V1 to V2 The amount of work done by the gas is greast, when the expansion is :...An ideal gas is filled in one part of a chamber having two parts. The other part is evacuated. If a hole is made in the ...Most accurate values are :-...