Chemical EquilibriumHard
Question
Given the reaction between 2 gases represented by A2 and B2 to give the compound AB(g).
A2(g) + B2(g) = 2AB(g)
At equilibrium, the concentration
of A2 = 3.0 × 10-3 M
of B2 = 4.2 × 10-3 M
of AB = 2.8 × 10-3 M
If the reaction takes place in a sealed vessel at 527oC, then the value of Kc will be:
A2(g) + B2(g) = 2AB(g)
At equilibrium, the concentration
of A2 = 3.0 × 10-3 M
of B2 = 4.2 × 10-3 M
of AB = 2.8 × 10-3 M
If the reaction takes place in a sealed vessel at 527oC, then the value of Kc will be:
Options
A.4.5
B.2.0
C.1.9
D.0.62
More Chemical Equilibrium Questions
When S in the form of S8 is heated at 900 K, the initial pressure of 1 atm falls by 30% at equilibrium. This is because ...For the following mechanism, P + Q PQ R at equilibrium is :[k represents rate constant]...Find the concentration of manomeric dichloroacetic acid in a CCl4 solution which contains 0.0129 g of the acid in 100 ml...One mole of NaO4(g) at300 K is kept in a closed container under one atmosphere. It is heated to 600 K when 20% by mass o...The equilibrium constant of the following are : N2 + 3H2 ⇌ 2NH3            K1 N2 + O2 ⇌ 2NO      Â...
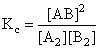
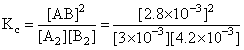 = 0.62
= 0.62