Chemical EquilibriumHard
Question
One mole of NaO4(g) at300 K is kept in a closed container under one atmosphere. It is heated to 600 K when 20% by mass of decomposes to NO2 (G). The resultant pressure is : NaO4(g)
Options
A.1.2 atm
B.2.4 atm
C.2.0 atm
D.1.0 atm
Solution
1 mole N2O4(i.e.,92g) is lept at 1 atm P and 300
K in a closed container, thus
P2 = 1 atm V = ? n = 1 mole,
R = 0.0821 L. atm. mol -1K -1, T = 300K
V =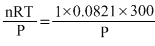 = 24.63L
= 24.63L
Now, N2O2 decomposes as
N2O2 ⇋ 2NO2
and 20% N2O4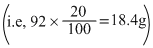
decomposes to NO2, thus
moles of NO2 formed = 2 × moles of N2O4
decomposed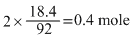
and moles of N2O4 remaining =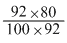
= 0.8 mole
and total moles of N2O4 and NO2 = 0.8 + 0.4
= 1.2 moles
Now P2 =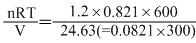
= 2 . 4 atm.
K in a closed container, thus
P2 = 1 atm V = ? n = 1 mole,
R = 0.0821 L. atm. mol -1K -1, T = 300K
V =
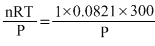 = 24.63L
= 24.63LNow, N2O2 decomposes as
N2O2 ⇋ 2NO2
and 20% N2O4
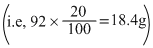
decomposes to NO2, thus
moles of NO2 formed = 2 × moles of N2O4
decomposed
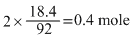
and moles of N2O4 remaining =
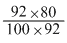
= 0.8 mole
and total moles of N2O4 and NO2 = 0.8 + 0.4
= 1.2 moles
Now P2 =
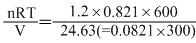
= 2 . 4 atm.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
At total pressure P1 atm and P2 atm, N2O4 is dissociated to an extent of 33.33% and 50.00%, respectively. The ratio of p...In the equilibrium mixture H2(g) + I2(g) $\rightleftharpoons$2HI(g), the mole ratio of gases are $\sqrt{2}:\sqrt{2}:10$,...The equilibrium $\text{SO}_2\text{Cl}_2(g) \rightleftharpoons \text{SO}_2(g) + \text{Cl}_2(g)$ is attained at $25^\circ\...For the reaction 2NO2(g) ⇋ 2NO(g) + O2(g), (Kc = 1.8 × 10-6 at 184oC) (R = 0.0831 kJ / (mol.K) When KP and Kc...The equilibrium constant for the reaction CO(g) + H2O(g) $\rightleftharpoons$CO2(g) + H2(g) is 3.0 at 500 K. In a 2.0 L ...