Gaseous StateHard
Question
A gas described by van der Waal′s equation
Options
A.behaves similar to an ideal gas in the limit of large molar volumes
B.has the pressure that is lower than the pressure exerted by the same gas behaving ideally
C.is characterized by van der Waal′s coefficients that are dependent on the identify of the gas but are independent of the temperature
D.behaves similar to an ideal gas in the limit of large pressures
Solution
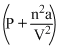 (V - nb) = nRT
(V - nb) = nRTAt low pressure, when the sample occupies a large volume, the molecules are so far apart for most of the time that the intermolecular forces play no significant role, and the gas behaves virtually perfectly.
a and b are characteristic of a gas and are independent of temperature. The term
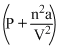 represents the pressure exerted by an ideal gas while P represents the pressure exerted by a real gas.
represents the pressure exerted by an ideal gas while P represents the pressure exerted by a real gas.Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Three footballs are respectively filled with nitrogen, hydrogen and helium. If the leaking of the gas occurs with time f...A gaseous mixture was prepared by takig equal moles of CO and N2. If the total pressure of the mixture was found 1 atmos...Which of following statement (s) is true I - Slope of isotherm at critical point is maximum. II - Larger is the value of...If most probable velocity is represented by ′a′ and fraction possessing it by ′f′, then with inc...Henry′s law constants for four gases are given below at 293 K. Which one is more soluble in water at that temperat...