Gaseous StateHard
Question
If most probable velocity is represented by ′a′ and fraction possessing it by ′f′, then with increase in temperature which one of the following is correct ?
Options
A.a increases, f decreases
B.a decreases, f increases
C.both a & f decrease
D.both a & f increase
Solution
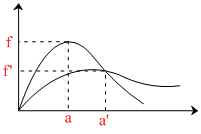
f′ < f
a′ > a
f = fraction of molecules posessing velocity (V)
a = most probable velocity.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Match of following (where Urms = root mean square speed, Uav = average speed, Ump = most probable speed) List I List II ...A gas cylinder containing cooling gas can withstand a pressure of 14.9 atmosphere. The pressure gauge of cylinder indica...A real gas obeying Vander Waal′s equation will resemble ideal gas, if the :...At 100oC and 1 atm if the density of the liquid water is 1.0 g cm-3 and that of water vapour is 0.0006 g cm-3 then the v...N2 + 3H2 → 2NH3. 1 mol N2 and 4 mol H2 are taken in 15 L flask at 27oC. After complete conversion of N2 into NH3, ...