Gaseous StateHard
Question
A gaseous mixture was prepared by takig equal moles of CO and N2. If the total pressure of the mixture was found 1 atmosphere, the partial pressure of the nitrogen (N2) in the mixture is
Options
A. 0.8 atm
B.0.9 atm
C.1 atm
D.0.5 atm
Solution
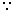 nCO = nN2
nCO = nN2∴ PCO = PN2
Given,PCO + PN2 = 1 atm
or 2PN2 = 1 atm
or PN2 = 0.5 atm
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Consider given reaction : Metal (M) Select correct statement(s)...Compressibility factor for H2 behaving as real gas is :...Two glass bulbs A and B are connected by a very small tube having a stop cock. Bulb A has a volume of 100 cm3 and contai...For gaseous state, if most probable speed is denoted by C*, average speed by and mean square speed by C, then for a larg...The average velocity of an ideal gas molecule at 27oC is 0.3/s. The avarage velocity at 927oC will be :...