ThermodynamicsHard
Question
At room temperature, the rms speed of the molecules of a certain diatomic gas is found to be 1930 m/s. The gas is
Options
A.H2
B.F2
C.O2
D.Cl2
Solution
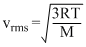
Room temperature T
 300 K
300 K∴

∴ M = 2.0 g / mol or the gas is H2
∴ Correct option is (a).
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
When you make ice cubes, the entropy of water...An ideal gas is taken through a process where dQ = dU - dW. The specific heat of the ideal gas in this process is (dQ = ...The value of Cp for a diatomic gas is :...At constant pressure wokdone in changing the volume from V1 to V2 is :-...three rods of identical cross-sectional area and made from the same material from the sides of an isoscales traangles AB...