Chemical Kinetics and Nuclear ChemistryHard
Question
The rate law for a reaction between the substances A and B is given by Rate = k[A]n [B]m On doubling the concentration of A and halving the concentration of B, the ratio of the new rate to the earlier rate of the reaction will be as
Options
A.(m + n)
B.(n - m)
C.2(n - m)
D.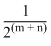
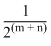
Solution
Rate1 = k [A]n [B]m; Rate2 = k[2A]n [1/2B]m
∴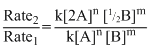 = [2]n [1/2m] = 2n.2-m = 2n-m
= [2]n [1/2m] = 2n.2-m = 2n-m
∴
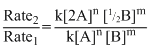 = [2]n [1/2m] = 2n.2-m = 2n-m
= [2]n [1/2m] = 2n.2-m = 2n-mCreate a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the first-order parallel reactions, starting with 1 M of A, which of the following statement(s) is/are correct? $A\o...For a process A+ B Products, the rate is first order with respect to A and First order with respect to B. When 1.0 mol e...The rate of a reaction doubles when its temperature changes from 300 K to 310 K. Activation energy of such a reaction wi...The bromination of acetone that occurs in acid solution is represented by CH3COCH3 (aq) + Br2 (aq) → CH3COCH2 Br (...Decrease in atomic number is observed during :...