SolutionHard
Question
Which of the following solution will have pH = 13 ?
Options
A.2g NaOH in 500 mL solution.
B.100 mL solution of 0.05 M Ca(OH)2.
C.100 mL solution 0.1 N Ca(OH)2.
D.4g NaOH in 500 mL solution.
Solution
a M = 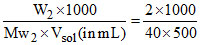 = 0.1 M
= 0.1 M
∴ [NaOH] = [OH-] = 0.1 = 10-1 M
pOH = 1, pH = 13
b. 0.05 M Ca(OH)2 = 2 × 0.05 N Ca(OH)2
∴ [OH-] = 2 × 0.05 = 0.1 = 10-1 M
pOH = 1 pH = 13
c. 100 mL of 0.1 N Ca(OH)2,
∴ [OH-] = 0.1 N = 10-1 M
pOH = 1 pH = 13
d. M =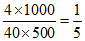 = 0.1 M
= 0.1 M
[OH-] = 0.2 M
pOH = -log[0.2] = -log 2 + log 10 = -0.3010 + 1 = 0.699
pH = 14 - 0.699 = 13.301
So the answer (a), (b) and (c) are correct.
∴ [NaOH] = [OH-] = 0.1 = 10-1 M
pOH = 1, pH = 13
b. 0.05 M Ca(OH)2 = 2 × 0.05 N Ca(OH)2
∴ [OH-] = 2 × 0.05 = 0.1 = 10-1 M
pOH = 1 pH = 13
c. 100 mL of 0.1 N Ca(OH)2,
∴ [OH-] = 0.1 N = 10-1 M
pOH = 1 pH = 13
d. M =
[OH-] = 0.2 M
pOH = -log[0.2] = -log 2 + log 10 = -0.3010 + 1 = 0.699
pH = 14 - 0.699 = 13.301
So the answer (a), (b) and (c) are correct.
Create a free account to view solution
View Solution FreeMore Solution Questions
For a dilute solution having molality m of a given solute in a solvent of mol.wt. M, b.pt. Tb and heat of vaporisation p...Y gm of non-volatile organic substance of molecular mass M is dissolved in 250 gm benzene. Molal elevation constant of b...If ′A′ contains 2% NaCl and is separated by a semipermeable membrane from ′B′ which contains 10%...On the basis of intermolecular forces predict the correct order of decreasing boiling points of the compounds:...A mixture contains 1 mole of volatile liquid A ($P_{A}^{o}$= 100 mm Hg) and 3 moles of volatile liquid B ($P_{B}^{o}$= 8...