Chemical BondingHard
Question
Which of the following are arranged in an increasing order of their bond strengths ?
Options
A.O2- < O2 < O2+ < O22-
B.O22- < O2- < O2 < O2+
C.O2- < O22- < O2 < O2+
D.O2+ < O2 < O2- < O22-
Solution
According to bond order values the given order is the answer. Bond order values are + 1, + 1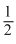 , + 2 and + 2
, + 2 and + 2 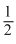 , higher bond order means stronger bond.
, higher bond order means stronger bond.
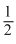 , + 2 and + 2
, + 2 and + 2 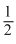 , higher bond order means stronger bond.
, higher bond order means stronger bond.Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following is the correct order of C = C bond lengths among these compounds ?I. CH3O - CH = CH – NO2 II. C...Predict the correct order among the following:...Carbon tetrachloride has no net dipole moment because of :...Find Ecell for the following cellMg(s)| Mg+2 (0.10 M)|| Ag+ (0.001 M)|Ag(s)Given : Eocell = 3.17 V = 0.06...Magnetic moment 2.83 BM is given by which of the following ions ? (At. nos. Ti = 22, Cr = 24, Mn = 25, Ni = 28)...