Chemical BondingHard
Question
Carbon tetrachloride has no net dipole moment because of :
Options
A.its planar structure
B.its regular tetrahedral structure
C.similar sizes of carbon and chlorine atoms
D.similar electron affinities of carbon and chlorine
Solution
due regular, symmetrical structure, μR = 0 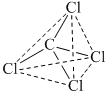

Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Match the following graphs :-GraphA (i) (+ deviation)B (ii) IdealC (iii) (-) deviation...Which of the following is correct :-...The helical structure of proteins is stabilised by:-...The type of hybrid orbitals used by the chlorine atom in ClO2- is :...In which of the following molecules/ions are all the bonds not equal?...