Chemical BondingHard
Question
Find Ecell for the following cell
Mg(s)| Mg+2 (0.10 M)|| Ag+ (0.001 M)|Ag(s)
Given : Eocell = 3.17 V
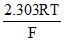 = 0.06
= 0.06
Mg(s)| Mg+2 (0.10 M)|| Ag+ (0.001 M)|Ag(s)
Given : Eocell = 3.17 V
Options
A.3.17 V
B.3.02 V
C.3.32 V
D.None of these
Solution
NCERT XII part 1 ; Page No. 71
Mg(s) → Mg+2 + 2e- (at anode)
2Ag+ + 2e- → 2Ag(s) (at cathode)
Mg(s) + 2Ag+ → Mg+2 + 2Ag(s)
Q =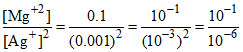 = 105
= 105
Ecell = Eocell - log10105
log10105
Ecell = 3.17 - × 5log1010 = 3.17 - 0.03 × 5
× 5log1010 = 3.17 - 0.03 × 5
= 3.17 - 0.15
= 3.02 V
Mg(s) → Mg+2 + 2e- (at anode)
2Ag+ + 2e- → 2Ag(s) (at cathode)
Mg(s) + 2Ag+ → Mg+2 + 2Ag(s)
Q =
Ecell = Eocell -
Ecell = 3.17 -
= 3.17 - 0.15
= 3.02 V
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Under ambient conditions, the total number of gases released as products in the final step of the reaction scheme shown ...The increasing order of the ionic radii of the given isoelectronic species is :-...The intermolecular interaction that is dependent on the inverse cube of distance between the molecules is :-...The pair of compounds that can exist together is...Pair of species among the following having same bond order as well as paramagnetic character will be-...