AIPMT | 2014Chemical BondingHard
Question
Magnetic moment 2.83 BM is given by which of the following ions ?
(At. nos. Ti = 22, Cr = 24, Mn = 25, Ni = 28)
(At. nos. Ti = 22, Cr = 24, Mn = 25, Ni = 28)
Options
A.Ti3+
B.Ni2+
C.Cr3+
D.Mn2+
Solution
μ = 2.83 BM, unpaired electrons = 2
28Ni+2 = 3d8 =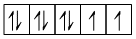
28Ni+2 = 3d8 =
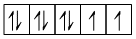
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
In which of the following pairs of molecules/ions, both the species are not likely to exist ?...When the value of (n + l) is not more than 3 which of the following subshells is not possible to exist :-...Consider the following species: CN+, CN–, NO and CN Which one of these will have the highest bond order?...The specific heat capacities of three metals are given below If 1.00 gm of each metal is heated to 100oC and added to 10...Which is not correct order of given property :...