Ionic EquilibriumHard
Question
The molar solubility in terms of solubility product Ksp is given by the relation
Options
A.s = 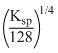
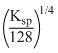
B.s = 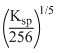
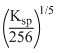
C.s = (256Ksp)1/5
D.s = (128Ksp)1/4
Solution
Ksp = 108s5
1 × 44 × s1+4 = 256 s5 = Ksp
1 × 44 × s1+4 = 256 s5 = Ksp
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The dissociation constant of a weak monoprotic acid is numerically equal to the dissociation constant of its conjugate b...The solubility of AgCl in water is 0.001435 g per litre at 15o C. The solubility product of AgCl is(Ag = 108, Cl = 35.3)...The phenomenon of optical activity will be shown by :-...The solubility products of Al(OH)3 and Zn(OH)2 are 8.5 × 10-23 and 1.8 × 10-14 respectively. If NH4OH is added...A weak acid (or base) is titrated against a strong base (or acid), volume v of strong base (or acid) is plotted against ...