Ionic EquilibriumHard
Question
The phenomenon of optical activity will be shown by :-
Options
A.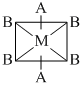

B.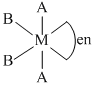

C.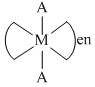

D.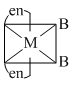

More Ionic Equilibrium Questions
The volume of the water needed to dissolve 1 g of BaSO4 (KSP = 1.1 × 10-10) at 25oC is:...The pH of a solution obtained by mixing 50 ml of 0.4 N HCl and 50 ml of 0.2 N NaOH is :...Which one of the following substances has the highest proton affinity?...What is the conjugate base of OH- ?...What is the minimum mass of NaBr which should be added in 200 ml of 0.0004 M-AgNO3 solution just to start the precipitat...